Journal of Clinical Investigation – First joint paper of new GvH-GvL Consortium
A new study lead by Prof. Kai Hildner from Erlangen University identifies BATF-expressing T cells as key players in intestinal acute graft-versus-host disease (GvHD).
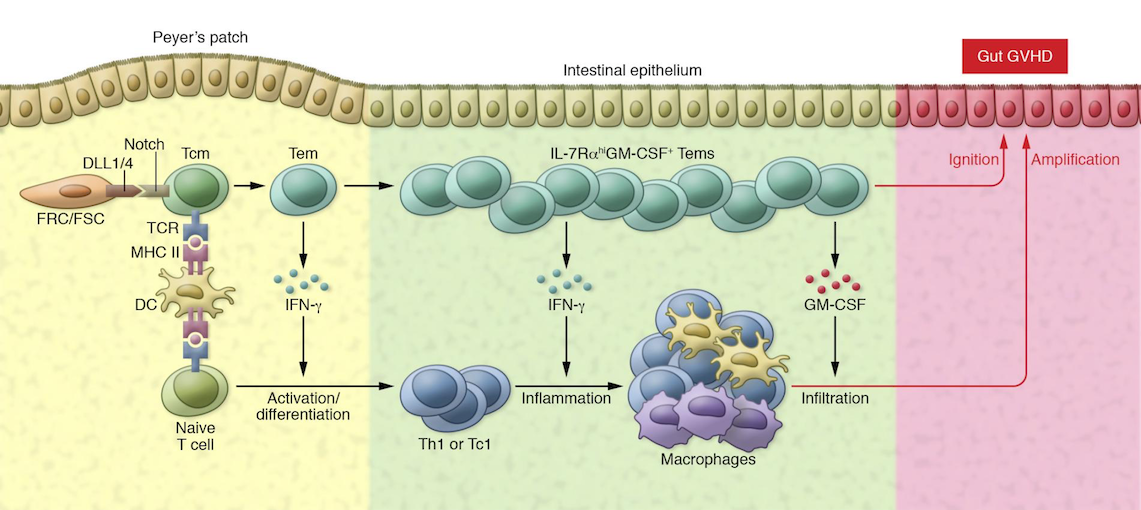
Alloreactive BATF+ effector memory T cells enhance inflammation by producing GM-CSF and augmenting the infiltration of innate immune cells in the intestinal mucosa. Zeng D, J Clin Invest 2018, © JCI.
Surprisingly, the transcription factor BATF controlled the formation of a distinct IL-7 receptor signaling–responsive GM-CSF+ T cell subset. These newly identified pathologic IL-7RhiGM-CSF+ T cells link IL-7, a cytokine previously mostly associated with T cell reconstitution, to the occurrence of acute GVHD. Furthermore, the study revealed GM-CSF derived from alloreactive T cells as a major driver of systemic and intestinal GvHD. These findings pinpoint the newly identified IL-7R/BATF/GM-CSF axis as a promising therapeutic target to prevent or even treat acute GvHD in patients undergoing allogeneic hematopoietic cell transplantation.
The new research article by Evelyn Ullrich et al. appeared today in the Journal of Clinical Investigation. The work resulted from a collaboration of an interdisciplinary team comprising several members of our newly founded DFG research consortium TR221 GvH-GvL between the partner universities Erlangen, Regensburg and Würzburg.
Further reading:
Commentary by Prof. Defu Zheng on this article in the Journal of Clinical Investigation.
Link to research article:








 From October 11th and 12th 2017 the annual international research conference organized by PhD students for PhD students of the
From October 11th and 12th 2017 the annual international research conference organized by PhD students for PhD students of the 