A gala event for cancer research
In an outstanding gala event, our joint research endeavor with the team of Prof. Harald Wajant was awarded to advance cancer immunotherapy.
 On November 16, 2019, the foundation Forschung Hilft! invited to the new Hotel Melchior Park in Würzburg for a fabulous fund-raising gala evening “Art & Music & Magic.” At the same event, our research effort towards developing new concepts for cancer therapy was honored: The foundation “Forschung Hilft” selected our research project for its annual Großen Förderpreis worth 20.000 Euros. Receiving this award was a wonderful and highly motivating experience knowing that Ms. Gabriele Nelkenstock and her dedicated team of the foundation raised the funds for this cancer research award from patients and committed private donors.
On November 16, 2019, the foundation Forschung Hilft! invited to the new Hotel Melchior Park in Würzburg for a fabulous fund-raising gala evening “Art & Music & Magic.” At the same event, our research effort towards developing new concepts for cancer therapy was honored: The foundation “Forschung Hilft” selected our research project for its annual Großen Förderpreis worth 20.000 Euros. Receiving this award was a wonderful and highly motivating experience knowing that Ms. Gabriele Nelkenstock and her dedicated team of the foundation raised the funds for this cancer research award from patients and committed private donors.
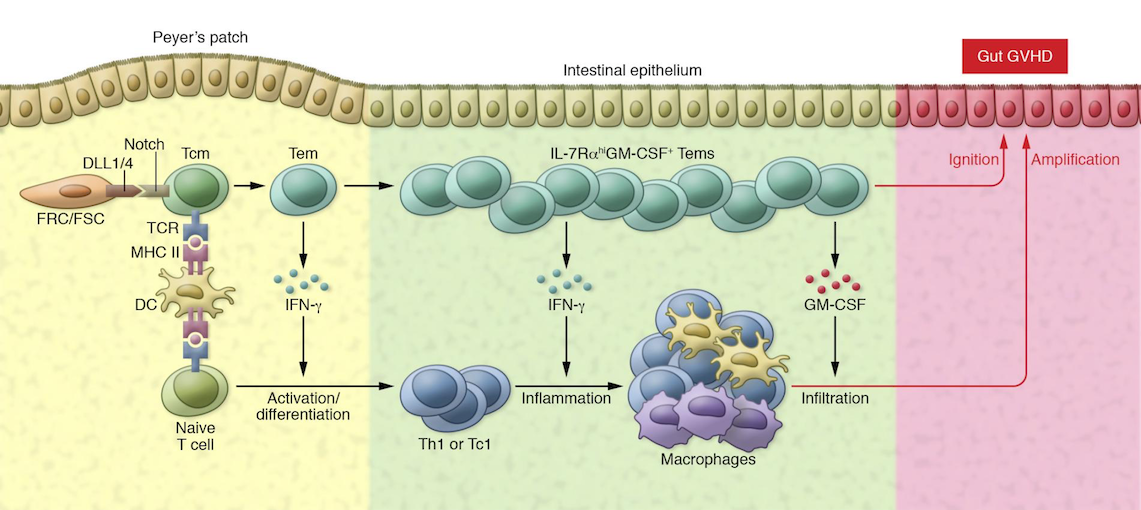
The award will directly support one of our cutting-edge projects to reactivate the body’s immune response against cancer. We want to achieve this by newly designed antibody fusion proteins. The effect of these antibody fusion proteins only unfolds in the immediate vicinity of cancer cells, so that the body’s immune response is only activated there. The advantage of our therapy concept is that no immune cells need to be manipulated from the outside. Furthermore, disadvantages of previously clinically tested immune-activating antibodies can be minimized, such as side effect profiles or systemic effects.
The research groups of Prof. Andreas Beilhack and Prof. Harald Wajant have collaborated on the immune regulation of inflammatory diseases and cancer for more than 10 years. In this collaborations, they particularly focus on the molecular and cellular mechanisms of action of the tumor necrosis factor receptor superfamily. The molecular biological and biochemical expertise of Prof. Wajant’s group and the experience of Prof. Beilhack’s group in immunotherapy and cancer complement each other in an excellent way. The interdisciplinary team consists of experienced biologists, molecular biologists, biochemists, bioinformaticians, technical assistants and clinicians.
The Große Förderpreis of the Foundation “Forschung Hilft!” sets the first foundation stone to develop a new type of cancer therapy. Thank you!


 The annual meeting of the immunology programs from Würzburg, Erlangen and Tübingen Universities took place in Obertrubach, Franconia. This year’s meeting was organized by Hans-Martin Jäck and his team from the University of Erlangen.
The annual meeting of the immunology programs from Würzburg, Erlangen and Tübingen Universities took place in Obertrubach, Franconia. This year’s meeting was organized by Hans-Martin Jäck and his team from the University of Erlangen.